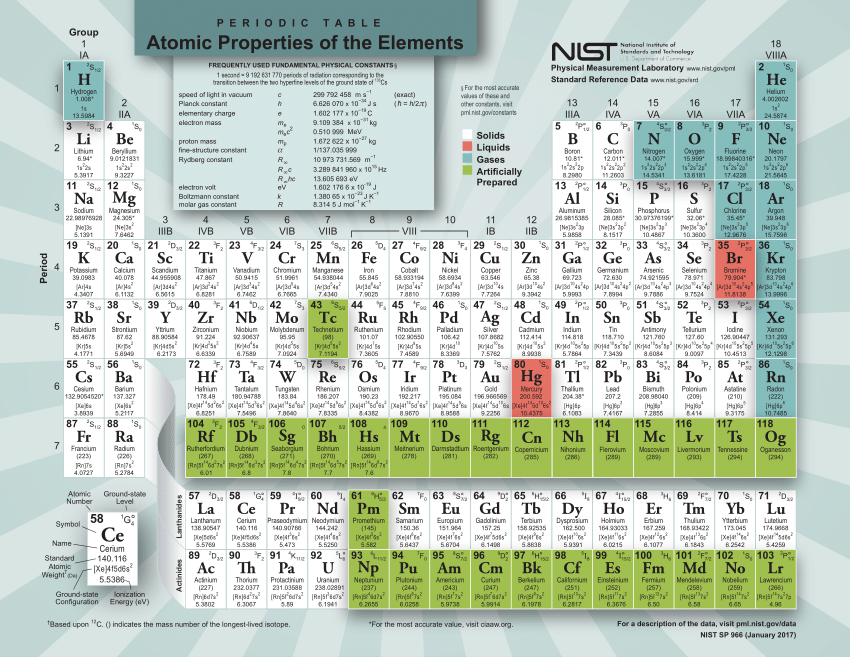 
All important information about the elements: Atomic number, valence electrons, oxidation state, electronegativity according to Allred-Rochow and Pauling, atomic mass, boiling point, melting point, atomic radius, density, history, discoverer, classification, crystalline structure type, electron configuration, basic state, ionization energy, isotopic composition, state of matter, hardness according to Mohs, oxidation numbers, percentage of mass in Earth's crust, year of discovery, half-life, and much more.Access the same amount of information on your browser, faster and larger than ever before with the same renowned ease of use.ĭownload now, or access directly online, free of charge and start experimenting. Our smartphone app (with over 4.89 million downloads!) is now joined by our newest update which features an online web version of our famous PTE smartphone app. Get informed with our mobile reference work, any time, with ease, offline (smartphone app) and in detail. Our app is a must-have of digital periodic tables. Later, after heating, the molecule releases a molecule of carbon dioxide (CO 2).The Merck KGaA, Darmstadt, Germany PTE App is the ultimate tool for every friend of chemistry - whether pupil or teacher, student or professor, amateur or expert, hobbyist or technician. The organic molecule cyclobutadiene (C 4H 6) combines with propenoic acid (C 3H 4O 2) to form a molecule with the molecular formula of C 7H 10O 2. You have of some unknown transition metal which weighs. Nitrogen and hydrogen: Carbon Dioxide (CO 2) Example Problem Moreover, this means that of NH 3 is equal to of NH 3. For ammonia, we should therefore get a molecular mass of. The last and final step is to add up the two products we got from multiplying. So, now we will multiply these numbers by their corresponding atomic masses. 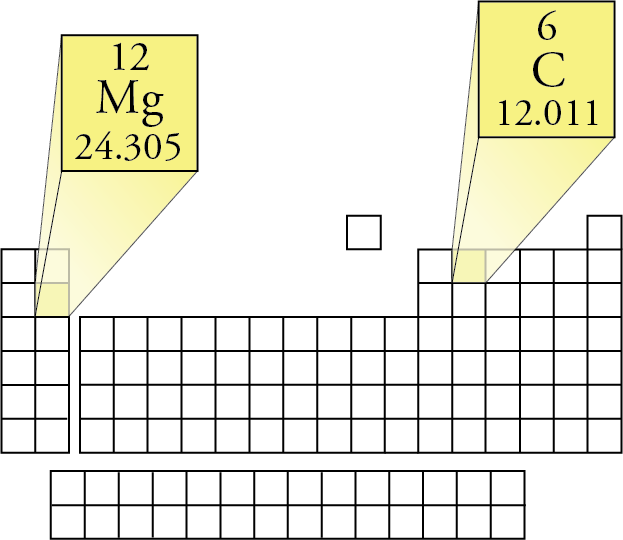
According to the molecular formula (NH 3), there is one nitrogen and three hydrogens present. The second step is to determine how much of each element is present in the compound. For example, if we are trying calculate for ammonia (NH 3), then we need to find the atomic masses for nitrogen and hydrogen. The atomic mass is equal to the atomic number which is listed below the element symbol. The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. Let’s look at some examples of calculating this value for some different molecules. It should be noted that this number is an average and therefore may vary due to isotopic elements. Molar mass can be calculated by using the periodic table and following three simple steps. The units of molar mass is grams per mole, often abbreviated. That’s a heck of a lot of molecules, which is why the molecular mass of table salt (sodium chloride) is a respectable 58.44 grams per mole, quite a large handful. Both definitions will give you the same result, they are just in different units.Ī “mole” of a substance is defined as atoms or molecules of that substance. 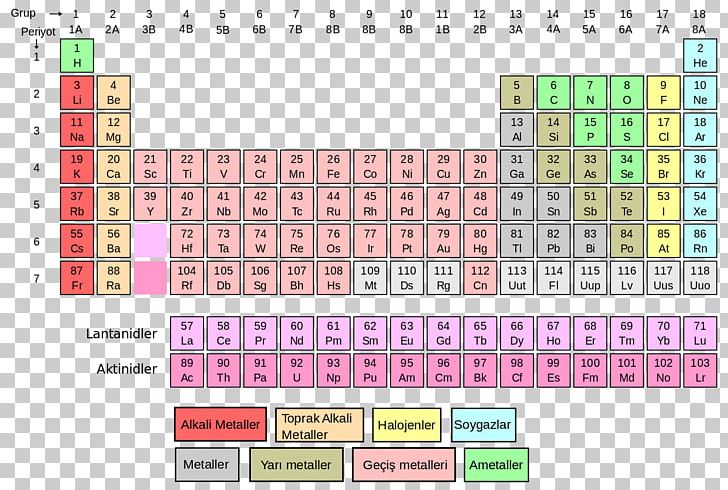
Another definition is the sum of the atomic weights of the atoms that make up a molecule. The definition of molar mass is simply the number of grams that one mole of a substance weighs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
